|
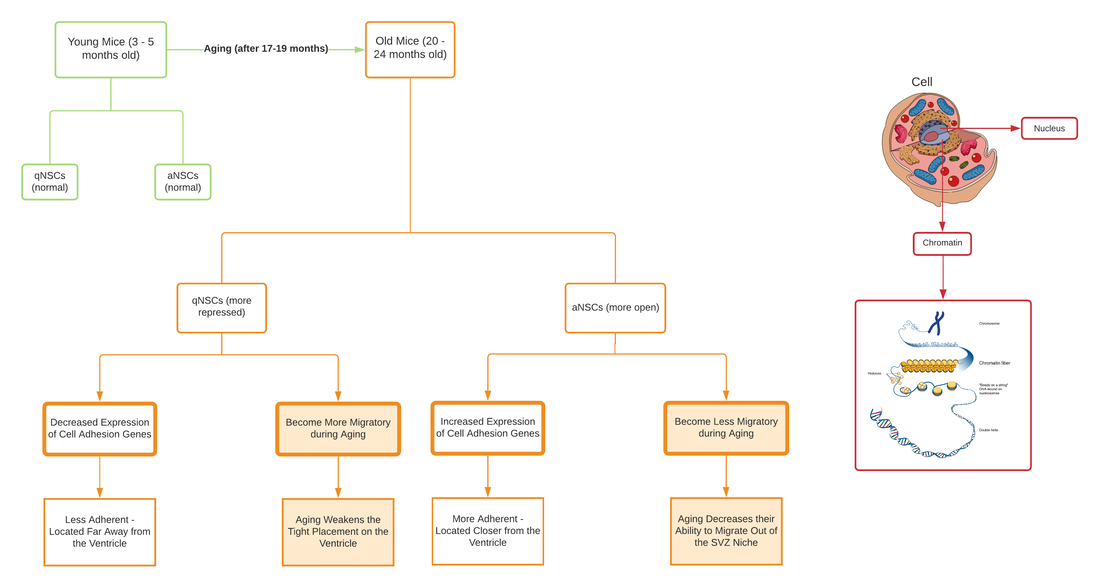
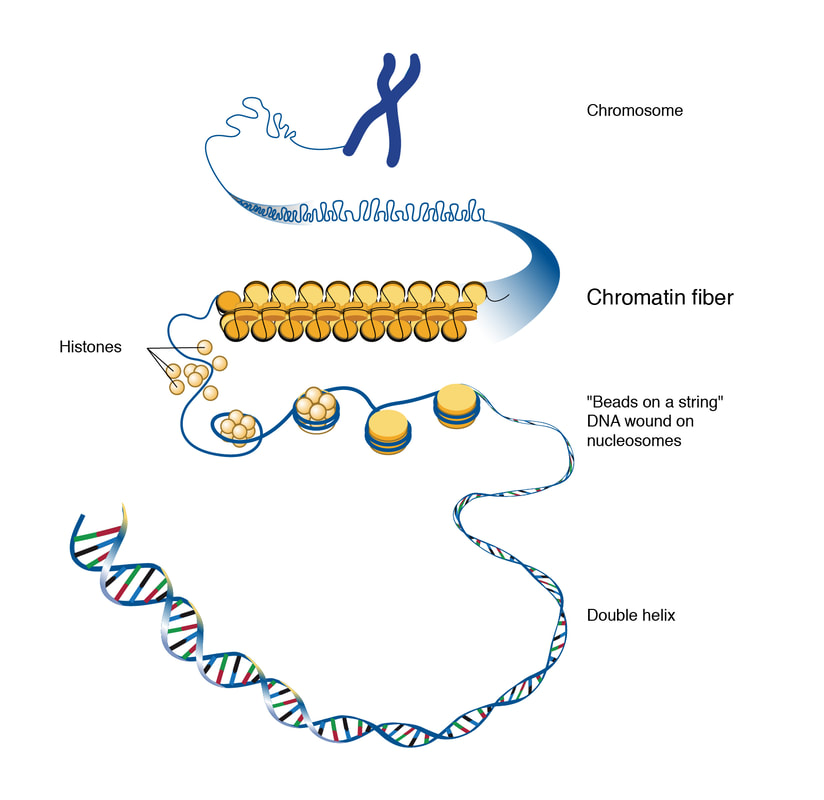
By Lucía Achucarro When we think about some of the biggest scientific questions that remain unsolved in the 21st century, we might think about Dark Matter, Quantum Entanglement, and even The Multiverse - concepts that the majority of us might feel distanced from on a daily basis. But what if I told you that amongst those abstract ideas, something as natural and familiar to us as aging can be found on that list? To this day, aging remains one of the biggest mysteries in science. As it turns out, aging is not as straightforward as we once thought, with many scientists branding it a “quirk of evolution”(1). Researchers explained that following the theory of natural selection (in which the more helpful a trait is at ensuring successful reproduction, the more it will be passed on), individuals with traits that prevent aging should have more opportunities than shorter-lived individuals to pass on their anti-aging genes – eventually giving rise to a whole population that lives longer and longer. As we know, that has not been the case. The uncertainty about how and why aging occurs has crucial repercussions for the world of science; aging is essentially a new frontier only beginning to be understood by the research community. Dr. Anne Brunet is one of the scientists at the forefront of research on aging. In her own words, “aging is an extraordinarily complex process that defies many conventional rules in biology” (2). Her work aims to dissect that process to truly understand the mechanisms behind it and thus come up with the most effective way to treat age-related issues in humans, especially for elderly people. As you can imagine, this task is no simple feat, which is why it requires someone with the utmost ability and determination - someone like Anne Brunet. Dr. Brunet’s Beginnings Born and raised in the commune of Bellegarde-sur-Valserine, France, Anne Brunet graduated in 1992 with an undergraduate degree in Molecular Biology at the Ecole Normale Supérieure, Paris. Afterwards she went on to obtain her Ph.D. in Cell Biology at the University of Nice, adding “Dr.” to her title in 1997. Her next step was to complete a postdoctoral fellowship in Neuroscience by 2003 at Harvard Medical School’s Radcliffe Institute(3), where she researched the molecular mechanisms that control an organism’s longevity. After considerable experience in her field, Dr. Brunet set her definitive sights on neuroimmunology and focused her main interest in understanding the mechanisms of aging, longevity, and the role of longevity genes in neural stem cells. Commenting on the topic and why she chose it, she stated, “aging is a wide-open question that has a lot of societal but also scientific implications, so I think it’s a really fascinating question”(4). Not only did she become an incredibly knowledgeable scientist, she was also extremely productive. With over 80 published papers and multiple awards, she set herself apart as a pioneer on the new scientific frontier of aging(5). Nowadays, Dr. Brunet centers the majority of her work at Stanford University where she applies her extensive knowledge in both teaching and researching as a Professor of Genetics and head of the Brunet Lab. The implications of the lab’s work go beyond understanding how aging works. Old age is a major risk factor in a number of diseases. By understanding aging, scientists can take significant steps towards formulating new and appropriate treatments for those diseases. Laboratory Models To figure out how aging works, Brunet’s research group has adopted a highly effective approach: using short-lived organisms as model systems. The organisms they currently study are Caenorhabditis elegans (a type of invertebrate nematode or roundworm), Nothobranchius furzeri (also known as the African killifish), and the common house mouse (Mus musculus) (2). The reason these organisms are so advantageous for their studies is that they age quickly: in the laboratory, C. elegans has an average lifespan of 18 to 20 days (6), while N. furzeri has an average lifespan of 12 weeks (7). Mice also have a relatively short lifespan (1-3 years), but have the added benefit of being mammals. This means that any findings in mice can be more easily applied to understand the same processes in humans, which are more closely related to mice than the other two species. The researchers are also not limited to studying aging in a whole organism; they conduct experiments on human cells as well, especially regenerative stem cells. This means that the researchers are able to study the effects of aging in a shorter period of time, yielding results in a quick and practical way. Additionally, all of their projects focus on understanding aging through different perspectives, including how aging is affected by diet and metabolism, the effects of aging on the brain, and even the possibility of suspended animation in humans (the temporary slowing or stopping of biological functions to preserve physiological capabilities). The Early Steps Towards a Cure for Aging-Related Defects Let’s take a closer look into one of the lab’s most recent publications: Chromatin accessibility dynamics of neurogenic niche cells reveal a reversible decline in neural stem cell migration during aging (Yeo et al. 2021, bioRxiv) (8). This paper found new ways in which defects in the brain - caused by aging or brain injury - can be studied and prevented. Don’t worry if some of the scientific terms or concepts look unfamiliar to you – let's begin with a breakdown of what they mean. Stem cells, from which all cells with specialized functions are produced, are also found in the brain. It is common knowledge that the regeneration and repair potential of the stem cell areas in the brain deteriorate as we age. The average adult brain contains neural stem cells present in two stages: Quiescent Neural Stem Cells (qNSCs) and Activated Neural Stem Cells (aNSCs). Quiescent Neural Stem Cells can become activated (aNSCs) and generate Neural Progenitors (NPCs) and Neuroblasts that can give rise to new neurons and glial cells, regenerating any part of the brain that needs it. It is this ability to regenerate that decreases as we age. Another term used frequently in the paper is Chromatin. Chromatin is the material that makes up a chromosome and it is a complex of DNA and protein (9). The proteins in chromatin, called histones, help package the yard-long DNA compactly like a coil, as shown in the flowchart below, so that it fits inside of the cell’s very tiny nucleus. The nucleus, located in the center of the cell, is not even 10 micron in diameter (9). The key point is that any changes in the chromatin structure are due to DNA replication and/or gene expression because these processes require chromatin to open up. So according to this, we can confidently infer that the chromatin structure changes as we age.
Let’s delve deeper into what this means, starting with how it affects cell adhesion. Cell adhesion is the process by which cells interact and attach to each other through special molecules on the cell surface; it plays a crucial role in cell communication and regulation and in the development of tissues. In the study, the chromatin profiles of the isolated cells revealed that in qNSCs and aNSCs, aging leads to opposing changes in the mRNA expression of genes involved in cell adhesion: old qNSCs have a decreased expression of said genes while old aNSCs display an increased expression of the same genes. To examine this effect further, they stained the coronal brain sections of the young and old mice with antibodies to determine the location of qNSCs and aNSCs in vivo (directly in the living organism). The researchers found out that in old brains, qNSCs are located further away from the ventricle and become less adherent to it upon aging, while aNSCs are located closer to the ventricle and become more adherent to it upon aging. These results support the claim that the adhesive properties of Neural Stem Cells change with age. Moving on, let’s analyze the effects on cell migration. Cell migration is the directed movement of cells in response to chemical or mechanical signals, it can occur during tissue renewal and repair. Continuing the experiment and once again using the chromatin profile combined with in vivo testing, researchers observed how qNSCs and aNSCs exhibit opposing changes in cell migration with age. The in vivo testing included injecting young and old mice with EdU, a chemical that integrates into the DNA of replicating cells so as to label and trace aNSCs and their progeny (NPCs). The experiment demonstrated that qNSCs become more migratory during aging, whereas aNSCs and their progeny become less migratory and lose the ability to be deployed as efficiently. These results sustain that aging weakens the tight placement qNSCs have on the ventricle and lessens the ability aNSCs have of migrating out of the SVZ niche. However, after observing and confirming this disadvantageous effect, the researchers hatched a solution involving the kinase ROCK. This specific kinase regulates actin cytoskeleton - a network that mediates, among other things, cell migration (10). To test their theory, they implemented a molecule inhibitor to target ROCK in cultured qNSCs and aNSCs. The authors observed how inhibiting ROCK in old aNSCs and progeny eliminated cell adhesive forces and, as a result, boosted their migration speed, revitalizing them. These results corroborate the theory that ROCK can be used as a molecular target to reverse the age-related decrease in aNSCs/NPCs migration. The summary presented here cannot begin to disclose the true complexity and sophistication of the study, but the outlined experiments and findings provided above are already enough to reflect its weight and scientific importance. In fact, this paper was chosen in particular because it’s the first one to identify changes caused by aging in the genome of the chromatin landscape of quiescent and activated neural stem cells. The potential that the above findings have is especially significant in the area of brain injuries and aging; this work has major implications in restoring the migratory ability and potential of NSCs during both cases. This study could also reveal new ways of restoring age-related defects in regeneration and repair by focusing on adhesive and migratory changes as people age, and its principles could help find ways to restore migration in the aging or injured brain. Dr. Brunet's Legacy Dr. Brunet’s contributions are incredibly valuable to all of us from multiple perspectives. For one, by taking a look at her work, girls and women all around can realize that they too can be at the front of their field if they choose to do so. Second, her research and leadership is helping us to make major headway towards understanding aging and its related diseases. Dr. Brunet’s work inspires confidence and faith that the human race is diligently moving forward towards understanding even the most complex mysteries that remain unsolved. References (1) Johannes Gutenberg Universitaet Mainz. “Why we did not evolve to live forever: Unveiling the mystery of why we age.” Science Daily, 15 September 2017, https://www.sciencedaily.com/releases/2017/09/170915144151.htm. Accessed 8 July 2021. (2) The Brunet Lab. “Research Interests.” The Brunet Lab at Stanford University, http://web.stanford.edu/group/brunet/research%20interests.html. Accessed 10 July 2021. (3) Harvard Radcliffe Institute. “Anne Brunet.” Harvard Radcliffe Institute, https://www.radcliffe.harvard.edu/people/anne-brunet. Accessed 27 July 2021. (4) Stanford University, creator. Stanford Faculty - Meet Anne Brunet. Stanford University, 2014. YouTube, https://www.youtube.com/watch?v=qjCApNPevVU. Accessed 1 July 2021. (5) Simons Foundation. “Anne Brunet, Ph.D.” Simons Foundation, https://www.simonsfoundation.org/people/anne-brunet/. Accessed 4 July 2021 (6) Zhang, Siwen, et al. “Caenorhabditis elegans as a Useful Model for Studying Aging Mutations.” Frontiers, 2020, https://www.frontiersin.org/articles/10.3389/fendo.2020.554994/full. Accessed 9 August 2021. (7) Valdesalici, Stefano, and Alessandro Cellerino. “Extremely short lifespan in the annual fish Nothobranchius furzeri.” PubMed, 2003, https://pubmed.ncbi.nlm.nih.gov/14667379/. Accessed 9 August 2021. (8) Yeo, Robin W., et al. “Chromatin accessibility dynamics of neurogenic niche cells reveal a reversible decline in neural stem cell migration during aging.” 2021, bioRxiv, p. 67 https://www.biorxiv.org/content/10.1101/2021.03.29.437585v1.full. Accessed 27 June 2021. (9) National Human Genome Research Institute. “Chromatin.” National Human Genome Research Institute, https://www.genome.gov/genetics-glossary/Chromatin. Accessed 10 July 2021. (10) Robertson, Sally. “What is the Actin Cytoskeleton?” News Medical Life Sciences, 2019, https://www.news-medical.net/life-sciences/What-is-the-Actin-Cytoskeleton.aspx. Accessed 4 July 2021.
|
Archives
February 2023
DiscovHER BlogScientista DiscovHER is a blog dedicated to discovHERies made by women in science. Follow us for links to the latest resHERch! Categories
All Alexandra Brumberg Amy Chan Avneet Soin Chemistry Diana Crow Engineering Health/medicine Indulekha Karunakaran Iqra Naveed Johanna Weker Lidiya Angelova Michael Clausen Mind Brain And Behavior Muhammad Hamza Waseem Nikarika Vattikonda Opinion Prishita Maheshwari-Aplin Technology Uma Chandrasekaran |
The Network for Pre-Professional Women in Science and Engineering
The Scientista Foundation is a registered 501(c)(3) -- Donate!



 RSS Feed
RSS Feed